Honeycrisp - some preliminary observations
Honeycrisp
is a new apple variety for New York that has created lots of market enthusiasm.
However, problems with this variety could destroy market confidence for the
apple. The most important postharvest problems are bitter pit and soft scald.
Bitter pit generally is thought to be related to calcium concentrations in
the fruit. Its incidence is often worse in younger, vigorously growing trees
than in mature, evenly cropping trees. Therefore, a combination of preharvest
calcium sprays together with good orchard management will likely result in
satisfactory control of pit. We are currently investigating the effectiveness
of calcium sprays for pit control and initial results should be available
by spring. Soft scald, however, has been of greater concern because its appearance
in the market place has been sporadic. Also, the problem is sometimes expressed
as an internal browning that is not apparent on the outside of the fruit.
In the 1999 harvest season, we carried out our first trials with Honeycrisp.
We focussed on the effects of:
1. Temperature, because soft scald may be a chilling-injury, and therefore
controlled by raising the storage temperature.
2. Diphenylamine (DPA) application, because this antioxidant compound, when
used for control of superficial scald, has been effective for control of soft
scald in other varieties.
3. Delays before storing fruit at low storage temperatures, as sometimes this
can control the disorder.
Fruit were harvested in a Western New York orchard on September 17, 1999,
and treated as described in Table 1. Soft scald was reduced by storing fruit
at 36oF compared with 33oF, and by treatment of fruit with DPA at harvest,
but these reductions in incidence, while significant, did not eliminate the
disorder. In contrast, a delay of a week at 50oF prior to either 33 or 36oF
storage markedly reduced soft scald incidence, overall averaging 0.6% compared
with 19% in the non-delay treatments. Factors such as firmness and soluble
solids were not affected by treatment, but the average bitter pit incidence
of fruit subjected to a delayed cooling treatment was 25% compared with 14%
in the non-delay treatment. Therefore, use of delayed cooling treatments should
be used only on fruit with a low bitter pit risk.
| Table 1: Soft scald incidence in Honeycrisp fruit untreated or treated at harvest with 1000ppm DPA, and then stored at 33o or 37oF, or kept at 50oF for one week before being stored at 33o or 36oF. | |
|
Treatment
|
Soft Scald (%)
|
|
Stored at 33oF
|
28a
|
| Stored at 36oF |
19b
|
| DPA-treated, stored at 33oF |
19b
|
| DPA-treated, stored at 36oF |
8c
|
| One week at 50oF, then stored at 33oF |
2cd
|
| One week at 50oF, then stored at 36oF |
0d
|
| DPA-treated, one week at 50oF, then stored at 33oF |
0d
|
| DPA-treated, one week at 50oF, then stored at 36oF |
0d
|
|
Values with different letters indicate
differences between means at the 5% level. |
|
We were reluctant to make recommenda-tions for use of delay
treatments for control of soft scald based on one year of data. However, trials
performed in 2000 have confirmed these results, and informal tasting of the
fruit does not suggest that Honeycrisp quality is affected by delayed cooling
treatments. Also, at least two growers have tried this or similar methods
with good results. Therefore, a tentative recommendation is made that a short
delay, either at 50oF or in a barn at similar temperatures, before cold storage
might be worth trying. In the longer term, however, storage temperature control,
perhaps 38-40oF, may be a better solution, at least for early to mid-harvested
fruit. This year's research, which is still in progress, suggests that a higher
storage temperature is less effective for late harvested fruit than early
harvested fruit. Further recommendations will be made available to the industry
before the 2001 harvest.
_________________________________________________________________________________________________
MCP - an update
1-Methylcyclopropene
or MCP is a new compound that will likely become available to fruit and vegetable
industries. It is an organic compound, which blocks ethylene receptors and
prevents ethylene effects in plant tissues for extended periods. MCP can have
major effects on ripening of apple fruit, and undoubtedly will have an enormous
impact on our apple industry as well as those elsewhere.
A summary of MCP research carried out at Cornell University can be found in
the New York Fruit Quarterly 8(3): 5-9 (2000), and will not be repeated here.
However, one issue of focus during the 1999 harvest season was a comparison
of effects of treating fruit warm and treatment of cold fruit on the day following
harvest using Cortland, Delicious, Empire, Gala, Jonagold, McIntosh, and Redcort
varieties. It is unclear at this stage what the optimum treatment conditions
for our apple varieties will be under commercial conditions. We observed three
types of response, and these are illustrated in Figure 1. Delicious responded
less well to MCP when treated cold than warm. Effects of MCP on Gala were
independent of treatment temperature. Jonagold responses were better for cold
fruit than warm fruit. Cortland, Empire, and McIntosh responses were similar
to that of Delicious. Redcort was similar to Jonagold. These data suggest
the possibility that treatment with MCP might be optimal when fruit are treated
warm on the day of harvest, at least for some varieties. During the 2000 harvest
season we studied the effects of application temperature and the types of
delay between harvest and treatment that might occur under commercial conditions.
In addition we have been able to carry out extensive semi-commercial trials
with MCP with the support of the New York Apple Association. Results will
be reported to the industry at the 2001 Storage Workshop at Ithaca. AgroFresh,
Inc. (Rohm and Haas Co.) is actively pursuing Federal registration of MCP.
Acute toxicity, mutagenicity and product chemistry studies indicate a favorable
toxicology profile. In addition, 1-MCP is structurally related to plant-containing
materials, is applied at extremely low dose levels, and there is no expectation
of measurable residues in food commodities. EPA registration was granted for
flowers in April, 1999 and is expected for postharvest use on fruits and vegetables
in 2001.
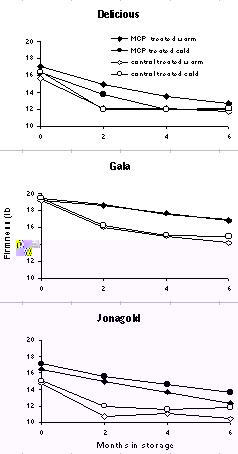 |
Figure 1 Flesh firmness of Delicious, Gala or Jonagold apples treated with 1ppm MCP either warm on the day of harvest or after cooling overnight and stored in air for up to 6 months. Evaluations were made after removal to 68oF for 7 days . |
________________________________________________________________________________________
Postharvest pathogens create new problems for apple storage
The two common postharvest
pathogens on apples are Penicillium expansum , the cause of blue mold, and
Botrytis cinerea, the cause of gray mold. These fungi have traditionally been
considered "wound pathogens", meaning that they invade fruit primarily through
wounds created during harvesting, packing, or transport. During the past 10
years, however, storage operators have noted increasing levels of decay in
unwounded fruit, especially Empire fruit, that are held in long-term CA storage.
Some of the results of our postharvest research were recently presented in
New York Fruit Quarterly 8(3): 24-28 (2000). Conclusions from the studies
reported there and from previous work can be summarized as follows:
1. The combination of DPA and benzimidazole fungicides (Benlate, Topsin-M,
Mertect 340F) provided excellent control of P. expansum and B. cinerea from
the early 1970's through the early 1990's.
2. Many storages now contain strains of P. expansum that are completely resistant to the DPA/benzimidazole treatment. Except for captan, no other fungicide is registered for postharvest use.
3. P. expansum can invade non-wounded Empire apples through the fruit stem during CA storage. Initial tests suggested that fruit with high boron content are more susceptible to stem invasion than are fruit with low to moderate levels of boron. Empire fruit are apparently more susceptible to stem invasion than are most other cultivars because most of the serious losses have occurred with Empire.
4. Huge numbers of spores of P. expansum can persist on empty bins that contained decayed fruit. Contaminated bins provide a venue for cycling inoculum from one season to the next. As the proportion of the P. expansum population that is resistant to fungicides increases, the level of decay increases and the inoculum load on bins increases correspondingly.
5. In the absence of new postharvest fungicides, improved decay control can only be achieved by improving packinghouse sanitation.
_____________________________________________________________________________________________________
Postharvest pathogens may be contributing to lost sales and
low prices
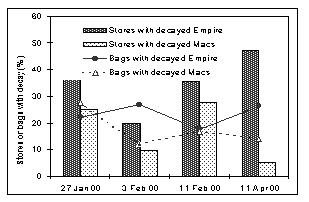
Decay Problems Evident in Retail Stores: A survey was conducted during winter of 2000 to determine if and how often rotten apples appeared in consumer packages presented for sale in chain stores. Data collectors for this survey visited 22 to 25 grocery stores in the mid-Hudson Valley (Newburgh, New Paltz, Kingston, Poughkeepsie) on each of four dates. Stores were not notified of the visits. The data collectors were instructed to peruse the displays of bagged apples as if they intended to make a purchase. They recorded the varieties of bagged apples on display and noted whether or not the bags contained any visibly decayed fruit. If decayed fruit were evident in one or more bags, the data collectors counted the number of bags on display and recorded how many of those bags contained decayed fruit. To avoid being conspicuous, data collectors did not examine every bag in a multi-layered display. Instead they attempted to record what consumers would see if they looked at the display with the intention of making a purchase. This method of checking for decays probably under estimated the actual levels of decay in bagged fruit because some decayed fruit may have been hidden within the bags.
Bagged Empire and McIntosh were available in at least 16 and sometimes in as many as 21 stores on the various survey dates. The proportion of Empire displays that contained decayed fruit varied from a low of 20% on February 3 to a high of 47% on April 11 (Fig. 2). Among those displays that contained decayed Empire fruit, the proportion of individual bags that contained decays ranged from 18% to 27%. For McIntosh, the proportion of stores with decayed fruit on display ranged from 10-28%, and 12-27% of the individual bags in those displays contained decays. In some cases, juice from decayed fruit had leaked through the bags leaving the surface of the plastic bags sticky with residue.
Figure 2. Percentage bagged apple displays in retail stores that contained decayed fruit, and estimated proportions of bags in those displays that contained decays. Results are based on surveys of 17-20 stores on each of four dates. The survey reported here was conducted in the Hudson Valley, but the decay problem is not limited to any single region. Labels on bags that contained decayed fruit showed that fruit originated from all of the various production areas within New York as well as from Massachusetts, Vermont, Pennsylvania, Washington State, and Ontario.
Editorial Opinion & Discussion Related to the Retail Store Survey: Many factors are contributing to the sorry condition of apples in retail stores. As noted earlier, we no longer have effective fungicides for controlling postharvest decays, and Empire has turned out to be a very decay-susceptible variety. Inoculum levels on bins and in packinghouses presumably have been increasing each year as the populations of fungicide-resistant strains of P. expansum have become predominant. As a result, apples going into bags are exposed to increasing concentrations of spores and are more likely to develop decays at wounds or stem punctures. Fruit to fruit contact in bags ensures that there will always be some freshly-wounded fruit.
Changes in chain store requirements for bagged apples are also contributing to decay problems. Instead of 3-lb bags, chains are now demanding 5-lb and 10-lb bags. With more fruit per bag, the probability increases that at least one fruit in the bag will develop decay even if the percentage of decay per 100 fruit remains the same. When fruit are packed in cells or on trays, decays can be individually removed and discarded at the retail level, but decayed fruit cannot be easily removed from bags. The move toward selling larger fruit in bags rather than in loose displays therefore increases the probability that the consumers will find decayed fruit on display.
Another significant factor contributing to decays in bagged fruit is the increasingly common demand by chain stores that bagged apples be packed in store-brand poly bags. The use of store-brand bags has created inventory nightmares for packing house operators. More importantly, store-brand bags are contributing to decay problems at the retail level because packinghouse operators can no longer move out packed inventory in a timely manner.
Most packing houses pack tray and/or cell packs of large-sized fruit at the same time that they are packing poly bags of smaller sized fruit. In many cases, orders for the larger (and often more profitable) cell-packs drive decisions concerning what varieties and what lots of fruit will be packed on any given day. The smaller fruit coming off of the line at the same time go into bags on the assumption that the broker will "find a home" for the bagged fruit within a few days. But what brand of bags should the storage operator use when a buyer has not yet been identified? When bagged apples were sold with the producer's or packer's name on the bag, the inventory of packed, poly-bagged fruit could be rotated fairly quickly because the packed inventory could be sold to a number of different retail chains. With the advent of store-brand poly bags, the bagged fruit can go to only one chain store. If that chain buys from a different broker or decides to feature Washington State apples for the next several weeks, the packed bags languish in cold storage while the fruit loses firmness and decay fungi begin to take their toll.
How can the industry address the marketing problem that exists when decayed fruit appear in poly bags? Solutions might include one or more of the following:
1. Improved sanitation in packinghouses might reduce the probability that fruit going into bags will develop decay. (No inoculum means no decay). Steps can be taken immediately, as outlined later in this report, to improve sanitation in packinghouses. However, considerable time, research, and facility modifications may be required before the industry can solve the current decay problem via sanitation alone.
2. Packing house operators could refuse to put fruit into bags until they know when and where the bagged fruit will be sold. However, it seems unlikely that anyone will pass up a potential sale of trays or cell packs just because they have not yet generated a sale for the bagged fruit that will come off of the packing line at the same time.
3. Larger, consolidated packinghouses and sales desks might allow more efficient handling of orders from the relatively few chain store buyers, thereby reducing situations where inventories of bagged fruit must be held for extended periods before they are sold. However, consolidation alone does not ensure that a better quality product will reach consumers.
4. The apple industry in New York, New England, and Michigan could band together and refuse to pack fruit in store- brand poly bags. Eliminating store-brand bags would cut packing costs at the same time that it would allow more orderly marketing of bagged fruit. However, historical precedent suggests that almost everyone in the apple industry would rather die than cooperate. The industry unity that would be needed to eliminate store-brand bags is not likely to be found in the near future.
Given the current status of the apple industry, none of the proposed solutions seem very practical. Without some action, however, decays in packed fruit will continue contributing to reduced consumption of apples and further declines in apple movement and prices.
____________________________________________________________________________________
Determining inoculum sources for postharvest decays
Relatively little is known about the sources of inoculum that contribute to postharvest decays caused by P. expansum. Inoculum can accumulate in recirculating postharvest drenches and in water flotation tanks, but where do these spores originate? For many years, pathologists believed that the primary source of Penicillium inoculum was soil carried on the bottoms of field bins. More recently, however, we began to suspect that inoculum recycles from year to year on the field bins themselves.
Inoculum carried on bins: To determine how many spores can be carried on bins, empty bins were "washed" using a portable postharvest drencher. Water samples were collected after each set of five bins had been washed, and inoculum levels in the water samples were determined by dilution plating. The number of spores recovered per bin was calculated by taking into account the number of bins washed, the total volume of the wash water in the drencher, and the number of colonies that developed per milliliter of water placed on dilution plates.
The bin-washing experiment showed that a single contaminated bin can carry more than two billion spores (Table 2). Although plastic bins carried only about one-fourth as many spores as the wooden bins from the same CA room, the plastic bins still harbored more spores than their relatively "clean" appearance would have suggested. Running 100 bins that each release 2 billion spores through a postharvest drencher that contains 1000 gallons of solution would raise inoculum concentrations in the drencher solution to more than 50,000 spores/ml. For many postharvest experiments with Empire fruit, spore concentrations of 20,000 spores/ml are enough to cause 100% decay in wounded fruit. Thus, inoculum carried on wooden bins in clearly sufficient to account for carry-over of inoculum from year to year.
A bin sanitation experiment was conducted during July 1999 to determine effectiveness of sanitizing bins with a quaternary ammonia solution (Deccosan 315). All decayed fruit were removed from empty wooden bins and the bins were then washed with Deccosan 315. This process resulted in a 99% reduction in the number of spores that could be retrieved from the bins, but some of the sanitized bins still carried nearly 8 million spores. The economic benefits of sanitizing bins remains unproven, but a 99% reduction in carry-over inoculum would seem to be a step in the right direction.
Will chlorinated water in the water dumps on packing lines sanitize bins? Apparently not: The bins from which we recovered 2 billion spores came from a packing line that fastidiously maintains chlorine levels in their water flumes. Thus, it appears that effective sanitizing of bins can only be completed by applying quaternary ammonium santiziers (or perhaps steam) in a separate operation after bins are emptied.
Table 2: Numbers of Penicillium spores per bin as determined by washing bins with a portable drencher and dilution-plating sub-samples from the wash water. Spores/bin Summer 1999 Summer 2000 Inoculum transported in air: Airborne spores in packinghouses are probably the major source of inoculum for contaminating apples after they come out of chlorinated water flumes on packing lines. Inoculum landing on bins after they come out of water dumps may also contribute to the total inoculum levels that we measured on bins.
A portable air sampler for agar plates (Burkard Manufacturing Co.) was used to measure concentrations of airborne Penicillium spores in packinghouses and storage rooms. Spore trapping was conducted at three packinghouses and their associated cold storage rooms on various dates during fall/winter of 1999-2000. In packinghouses, air was sampled close the water dump tank where apples are floated out of the bins and also at the far end of the packing line where automatic baggers are usually located. Air in cold storage rooms was measured by holding the spore trap about four feet above the floor in the middle of the storage room.
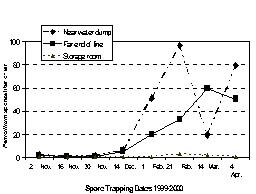
Concentrations of airborne inoculum were very low for November and December sampling dates, but spore concentrations near the water dumps approached 100 spores per liter of air by mid-February (Fig. 3). Agar plates exposed in still air adjacent to the spore trap showed that when spore concentrations approached 100 spores per liter of air as detected by the spore trap, then spores settled onto flat surfaces at the rate of approximately 2 spores per square inch per minute.
On each date, spore concentrations varied from one packinghouse to another, but all three locations showed the same pattern of low spore levels in the fall with increasing spore concentrations detected as the season progressed (Fig. 4). The decrease in spore levels detected near the water dumps on 14 March may have occurred because an unusual warm spell during the preceding week allowed storage operators to open overhead door for several days to take advantage of the warm spring air. Some of the accumulated spores in the packinghouses may have been exhausted when doors were left open for several days.
Figure 3. Number of Penicillium spores trapped at various locations in apple packinghouses. Data shown represents the means for three packinghouses on each date Spore concentrations in air were roughly half as great at the far end of packing lines as they were near the water dumps, and there were very few airborne spores in cold storage rooms (Fig. 3). Higher concentrations of spores near water dumps are expected because inoculum is brought in with bins and released as bins are lowered into the water dumps and as decayed apples meet brushes on the packing line. Levels of airborne inoculum in storage rooms remained low throughout the season despite the fact that the monitored storage rooms were "active" rooms with regular forklift traffic.
The levels of airborne inoculum detected in packinghouses suggest that anything coming out of the packinghouse during the mid-winter packing season will probably be contaminated with spores of P. expansum. Apples going into bags and packed boxes will carry inoculum that can cause decays while fruit are in transit to consumers. Empty bins can accumulate additional spores while awaiting removal from the packinghouse. Empty bins stored in CA rooms attached to the packing operation will be exposed to inoculum that drifts through the building from the packing line. The best method for reducing airborne inoculum in packinghouses has not been determined, but this aspect of sanitation deserves more study.
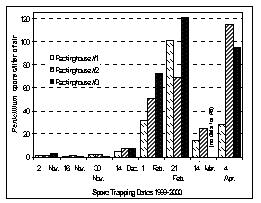
Figure 4. Numbers of Penicillium spores trapped near water dumps in three different packinghouses. ___________________________________________________________________________________
Getting Started with Packing House SanitationAlthough relatively little is known about the best sanitation methods for apple packing and storing operations, almost anyone can visualize improvements or changes that could help to reduce decay problems and subsequent inoculum levels. The following suggestions, in roughly priority order, are my "best bets" for cost-effective improvements in sanitation.
1. Chlorinate water flumes to kill fungal spores as fruit are immersed in water dumps. Of all the suggestions in this section, this is the only one that has been extensively tested. Chlorine in water forms hypochlorous acid, a very effective biocide that kills fungal spores on contact. However chlorine solutions do not penetrate decayed fruits and provide no residual activity to protect against future exposure to inoculum. Various sources of chlorine can be used to generate chlorinated water, but the most common sources are sodium hypochlorite and calcium hypochlorite. Household bleach is 5.25% sodium hypochlorite. Other more concentrated commercial formulations are also available.
Effectiveness of chlorine solutions varies depending on solution pH, concentration of free chlorine in the solution, solution temperature, and the duration of exposure. For best effect, the pH of chlorinated water solutions should be between 6.0 and 7.5. Lower pH's result in more rapid corrosion of equipment and more off-gassing of chlorine. The latter creates the "swimming pool" odor that can cause worker discomfort with prolonged exposure. At pH's above 7.5, the chemistry of the solution changes and biocidal activity is reduced.
The recommended concentration of free chlorine for water flumes in apple packing houses is 100 ppm. Lower concentrations may be adequate, but concentrations are likely to fluctuate considerably as organic matter settles into the flotation tanks and ties up the available chlorine. Therefore, water flumes should be charged with enough chlorine to bring free chlorine levels to 100 ppm at the beginning of each day and again at noon. Color test kits can be used to measure pH and chlorine concentrations. Swimming pool test kits often measure free chlorine only up to 5 ppm, but these test kits can still be used if flume water is diluted before it is tested. If one fluid ounce of chlorinated flume water is included in one quart of non-chlorinated water (= 1/32 dilution), then a concentration of 100 ppm in the flume water should produce the color reaction indicating 3 ppm free chlorine in the diluted sample. After several days of periodic testing, most packinghouse operators can derive a standard rate for charging with chlorine that will maintain 70-100 ppm free chlorine throughout the day.
Table 3: Effects of postharvest treatment on fruit firmness and on the incidence of decays caused by Penicillium expansum and Botrytis cinerea a in Empire fruit held in CA storages from 29 September 1999 through 14 July 2000. Number of decayed fruit per bin Fruit firmness 1P. expansum B. cinerea 14 Jul 21 Jul Storage A 23Storage B 1Fruit firmness was measured by testing both sides of 10 fruit per replicate when fruit were removed from cold storage on 14 Jul and again
after a 7-day shelf life test at 70oF.
2Postharvest treatments of DPA plus Mertect 340F applied by the packinghouse.
3Asterisks indicate that means are significantly different in the paired comparisons (P<0.05).Chlorinated water is appropriate only for water flumes. Chlorine cannot be used in combination with other postharvest treatments because chlorine cannot be combined in the same treatment tank with DPA or other fungicides.
2. Avoid postharvest treatment unless DPA is needed to control scald. In the absence of effective fungicides, postharvest treatment almost always results in increased incidence of blue-mold decay. Moving Empire fruit into CA storage without postharvest treatment should reduce the incidence of decay, and that in turn will reduce concentrations of airborne inoculum in storages and contamination problems on bins.
Postharvest treatment with DPA plus Mertect 340F is effective for controlling gray mold. Therefore, the incidence of gray mold may rise slightly when postharvest treatments are omitted. However, gray mold rarely causes the extensive losses in apples that frequently occur when fungicide-resistant P. expansum is distributed through bins via postharvest drenches.
The fact that postharvest treatments contribute to decay problems was verified in two trials conducted during 1999-2000 in cooperation with two growers and two Western NY packinghouses. For each orchard-storage combination, at eight bins of fruit were sent through the commercial drencher used by the packinghouse in fall of 1999 and another eight bins from the same orchard were moved into the same storage rooms without any treatment. The drenchers contained the usual mix of DPA plus Mertect 340F. Apples were removed from CA storage during early July of 2000, and the number of decays was determined by hand-grading 1000 fruit from each of the 8-bin treatments. The incidence of blue mold was consistently higher in fruit that had received a postharvest treatment (Table 3). In these experiments, the DPA-fungicide postharvest treatments did not result in improved fruit firmness.
3. Clean packinghouse floors regularly to remove inoculum. Spore trapping studies have shown that concentrations of airborne spores in packinghouses are very low early in the morning before there is any activity in the packinghouse. Spores from the previous day presumably settle to the floor when packing lines shut down in the evening and there is no longer any activity or air turbulence to help keep spores aloft. Wet mopping floors with an effective biocide (e.g., chlorinated water) before work starts each morning might significantly reduce spore concentrations in packinghouses.
4. Sanitize apple bins. Sanitizing apple bins will be difficult without purchase and installation of specialized equipment. "Rinsing" bins with a sanitizer such as a quaternary ammonium solution may help, but it will not be totally effective. Penicillium spores are hydrophobic (i.e., they repel water). Spores on blue-stained areas of bins are unlikely to be removed without some kind of brushing system, and this is true of plastic as well as wooden bins. A washer system that involved high-pressure sprays of a biocide combined with a brushing system would be ideal.
5. Devise methods to keep spores away from washed fruit. In the citrus industry where a different species of Penicillium that cause fruit rots, packinghouses are designed with walls separating the input and output ends of the packing lines. The fruit packing operation is physically separated from the part of the line where bins are emptied and decayed fruit are removed. The physical separation keeps spores from the "dirty" part of the packing line from blowing into the area where fruit are being packed.
Putting a wall through the middle of the apple packing line will be impractical in most situations. However, air flow patterns and locations of exhaust fans might be adjusted to minimize the movement of air from the water dump area toward the area where fruit are packed.
_____________________________________________________________________________________
What About Fungicide Alternatives?Captan used in postharvest treatments might prevent P. expansum from invading stems in Empire fruit. Captan has never been very effective for protecting against postharvest decays in wounds, but there is some evidence that it may be adequate for protecting stems. More tests are currently in progress. Where captan-treated fruit are acceptable to buyers, using captan in postharvest drenches might help to reduce decay in Empire fruit as compared to using only DPA plus Mertect 340F. However, avoiding all postharvest treatments on Empire fruit is still likely to be the best option.
Several biocontrol fungicides have been registered for postharvest use on apples, but they are either ineffective or unavailable due to distribution and marketing problems. In some cases, these products are only registered for use as line sprays and they therefore cannot be used in drenches prior to CA storage.
Novartis has a new fungicide with the trade name of "Scholar" that could prove useful as a postharvest treatment for apples. The generic name for Scholar is fludioxonil. It is a phenylpyrrole fungicide with a different mode of action than any of the other fungicides currently registered for field or postharvest use on apples. Scholar has been very effective against P. expansum in several trials conducted at the Hudson Valley Lab, but it probably will require at least two more years of testing before it can be registered.
Even if Scholar or some other new fungicide is eventually registered for apples, resistance to the new fungicide will develop quickly unless inoculum levels are kept to a minimum. The combination of DPA and benzimidazole fungicides remained effective for nearly 20 years only because of a fortuitous interaction between DPA and the fungicides that slowed development of resistance. In the absence of DPA, the benzimidazoles would have controlled postharvest decays for only about five years, and a similar fate can be anticipated for any other new fungicides that might be introduced unless the new product is managed carefully to forestall resistance.
Conclusions
Improved sanitation will be essential for minimizing losses during CA storage, for reducing the incidence of decayed fruit in consumer packages, and for preventing rapid development of fungicide resistant strains that would compromise any new fungicide that might be introduced. Basic sanitation measures that should be implemented immediately include the following:
1. Chlorinate water flumes in packing houses.
2. Do not apply postharvest fungicide/DPA treatments unless absolutely necessary.
3.Look for improved ways to sanitize floors and surfaces in packinghouses and to eliminate inoculum from contaminated bins.
_______________________________________________________________________________________
ReTain effects during storage of Empire apples
Last year we reported on CA storage of McIntosh fruit which had been untreated or treated with ReTain before harvest. In general, our results indicated that while effects of ReTain on fruit maturity, especially internal ethylene concentration, could be significant, no differences in storage quality were detected. In the1999 harvest season we carried out a similar experiment using Empire, a variety that produces less ethylene than McIntosh. Fruit were harvested from four Western New York orchards on two occasions. Fruit maturity was assessed on fruit from each orchard and then fruit from each orchard were stored under CA conditions (2% O2/2% CO2) at 36oF. Untreated and ReTain-treated fruit were stored separately, but for the first harvest date, separate CA chambers with mixed-treatments were also established. ReTain-treated and untreated fruit were mixed in the same chamber to determine if ethylene-production from the non-treated fruit would compromise the potential storage-enhancement that might be provided if Retain-treated fruit were never exposed to untreated fruit that were generating ethylene.
At the first harvest date, fruit from untreated trees in two orchards had high internal ethylene, but ReTain treatment prevented this increase (Table 4). By the second harvest on October 7, however, ReTain only controlled ethylene in orchard 4.
Fruit were removed from storage after 5 months and quality evaluated after 1 and 7 days at 68oF. ReTain-treated fruit were firmer than control fruit overall, but averaged only 0.3 lb greater firmness over both harvest dates. Because treatment effects for 1 and 7 days shelf life were the same, only data for 1 day of shelf life are shown (Table 5). These show that the response to ReTain during storage was greatly affected by orchard. There was no significant effect of treatment for orchards 1 to 3, but ReTain-treated fruit from orchard 4 was 0.9 lb firmer than the untreated fruit. The firmness benefit for fruit from this orchard was found at both harvests. Interestingly, orchard 4 had the most soft fruit at harvest (Table 4).
Untreated fruit had similar firmness on either day 1 or day 7, regardless of whether fruit were stored separately or with ReTain-treated fruit (Table 6). ReTain-treated fruit, however, were slightly firmer when stored with other treated fruit than with untreated fruit on day 1, but not on day 7.
Our conclusions are that ReTain treatment may result in significant maintenance of firmness, especially in fruit from an early maturing block. However, these effects are orchard-specific and inconsistent. In general, ReTain should be used as a harvest management tool rather than a means of maintaining quality during storage.
Table 4: Internal ethylene concentrations (ppm) and flesh firmness (lb) of untreated or ReTain-treated Empire fruit from four Western New York orchards harvested on two occasions. Harvest date Orchard # 9/28/99 1 2 3 4 10/7/99 1 2 3 4
Table 5: Flesh firmness (lb) of untreated or ReTain-treated Empire fruit from four Western New York orchards after 5 months of CA storage, evaluated after 1 day at 68 oF. Harvest date Orchard # 9/28/99 1 2 3 4 10/7/99 1 2 3 4
Table 6: Flesh firmness (lb) of Empire apples from four orchards in Western New York that were either untreated or sprayed preharvest with ReTain. Fruit were stored under CA conditions for 5 months, either separated or mixed, and tested after 1 or 7 days at 68oF. Storage type Separate Mixed _____________________________________________________________________________________
Ethrel effects on storage quality of McIntoshIn the Hudson Valley, spraying Ethrel on McIntosh trees to enhance fruit color and allow harvest before fruit drop occurs has become a standard management procedure for the industry. We are often asked about the effects of treatment on fruit quality, especially as some growers then store treated fruit under CA conditions. Ed Stover and I ran trials in 1996 involving storage of Ethrel -treated fruit.
Standard procedures used by the Hudson Valley growers were followed. These involved harvest of fruit within a 6-day window after Ethrel treatment (August 30). Fruit were transported to Ithaca, cooled overnight, and CA atmospheres applied. Fruit were assessed after 3 and 6 months, plus 1 or 7 days at 68oF.
Firmness of Ethrel -treated fruit harvested on September 4 was 14.3 lb compared with 15.0 lb in the untreated control fruit. Both untreated and treated fruit softened during storage for either 3 or 6 months, but Ethrel -treated fruit were always softer 1 day after removal (Table 7). Effects of treatment were small after a 7-day shelf life period. No effects of treatment on incidence of disorders were detected.
What does one conclude from these data? Ethrel-treatment permits harvest of fruit with marketable color before excessive fruit drop occurs. The present market puts a premium on color, rather than firmness, and in the absence of firmness standards this situation is likely to continue. However, our results suggest that Ethrel -treated McIntosh should not be held in CA more than 3 months. These data were obtained using fruit harvested at the latter part of the window for Ethrel use, i.e., 4-6 days after harvest. but depending on temperatures each season there will be considerable variation. Although the results are likely to generally applicable, slower responses might be expected from fruit treated with lower rates of Ethrel or harvested sooner after harvest. As a final comment, trials this year show that Ethrel-treated McIntosh will not respond to MCP.
Table 7 Flesh firmness (lb) of Hudson Valley-grown McIntosh apples either untreated or treated before harvest with Ethrel, and stored in CA for 3 and 6 months, pus 1 or 7 days at 68oF. Control Ethrel-treated ______________________________________________________________________________________
Acknowledgments:
The New York Apple Research and Development Program, the New York Apple Research Association, and Novartis funded the research reported here. Paul Woolley of Hilltop Nurseries provided the Honeycrisp fruit used for the 'delay' experiment. The ReTain experiments could not have been carried out without the help of Ken Silsby of Sun Orchard Fruit Company and their growers, and Regina Rickenburg of Valent BioSciences. We thank Fritz Meyer, Jackie Nock, Catherine Ahlers, and Albert Woelfersheim for excellent research assistance. Robert and Doug Minard provided the portable drencher used for washing bins to determine spores loads carried on bins. Thanks to Deborah Breth, Pete Russell, Rod Farrow, Sun Orchards, and Lake Ontario Fruit for cooperating on the trial comparing Empire stored with and without postharvest treatment. Thanks to Dressel Farms for providing CA storage for fruit involved in some of the experiments reported here.