

Genetic Engineering of Grapevines for Improved Disease Resistance
Julie R. Kikkert and Bruce I. Reisch. 1996. Genetic engineering of grapevines for improved disease resistance. In Grape Research News Vol. 7 No. 2, Summer, 1996. M. Goffinet (ed.). New York State Agricultural Experiment Station, Geneva, New York.
Department of Horticultural Sciences, New York State Agricultural Experiment Station, Geneva, NY 14456
Grapevine diseases cause growers to invest millions of dollars and numerous hours on various techniques to reduce losses. Grapevines with improved disease resistance would be welcomed, especially if other traits were not altered. Reduction of pesticide sprays by even one or two per year would cut the cost of production and may benefit the environment. The grapevine breeding program at the New York State Agricultural Experiment Station in Geneva is using both traditional breeding methods as well as biotechnology to develop disease resistant vines. This article focuses on the use of genetic engineering in which genes that code for desired traits are inserted into a plant. The major advantage of genetic engineering techniques is the ability to direct improvement of important cultivars without altering their essential features. Thus, we would like to develop a disease resistant 'Chardonnay' or 'Concord', for example.
Gene transfer technology became routine in the mid 1980's for easily manipulated non-woody plants such as tobacco. However, it has only been in the last few years that genetically transformed grapevines have been produced. This technology is now progressing rapidly, with at least 14 labs working worldwide to genetically engineer grapevines. Currently, transformed grape varieties are being tested in France and in the United States. Researchers collaborating between Kearneysville, West Virginia; Fresno, California; and Geneva, New York, are testing 'Thompson Seedless' vines carrying a gene for resistance to Tomato Ringspot Virus. In France, there are two groups testing rootstocks, as well as 'Chardonnay', with newly inserted genes for resistance to Fanleaf Virus, and a third group is testing Richter 110 with a gene for resistance to Chrome Mosaic Virus.
Genes that may confer disease resistance to plants are now available from a variety of sources. We have already mentioned the testing of virus resistance genes in grapevines. These genes come from a part of the virus itself. Resistance is based on the observation that once a plant becomes infected with certain viruses, it is resistant to future attacks. Thus, insertion of a non-infectious viral gene into a plant provides a sort of protective vaccine. Genes for resistance to fungi and bacteria work in different ways. Some genes that have been isolated from plants and higher fungi, code for enzymes (such as chitinase) that degrade a major component of the outer protective walls of certain fungi. Other genes act by creating holes in the membrane of fungal and bacterial cells. These membrane-active genes have been taken from a variety of organisms such as plants, mammals, amphibians, and insects. They have also been synthesized in the laboratory. Much work is needed to test the numerous genes against grapevine pathogens.
To successfully engineer disease resistance genes into plants, the following are needed: 1) recipient cells that are capable of growing into whole plants, 2) a method to transfer the genes into the cells, 3) proper expression of the genes by the transformed plant cells, 4) a method to select the transformed cells from the non-transformed cells, 5) regeneration of whole plants, and 6) evaluation of disease resistance. Success in grapevine transformation came only when researchers started using what are termed embryogenic cultures. These cultures are grown in the laboratory under sterile conditions in an artificial growth medium. The cultures consist of tiny clumps of cells that are capable of growing into embryos that can germinate into plants. The cells originate from the body of the plant (somatic cells) and not the egg or sperm cells, so that each embryo is a clone (exact replicate) of the original plant.
To insert genes into embryogenic cultures, most researchers working on grape transformation rely upon modified strains of Agrobacterium (the bacterium responsible for crown gall), which transfers genes into plants as part of its normal life cycle. In our Geneva laboratory, we have taken a different approach. We rely upon the biolistic process (short for biological ballistics), whereby DNA-coated particles of extremely minute size are used to carry foreign genes into grapevine cells. DNA coding for the genes of interest is coated onto the minute tungsten-microprojectiles. These are accelerated at extremely high speeds into the cultured cells using a biolistic device, also known as the "gene gun" (Figure 1). There are usually several genes transferred into each cell penetrated by a microprojectile. One of these genes might be the gene of interest coding for a desired trait. Another gene is used to help separate the transformed cells from the remaining normal cells. This is important because usually less than 5% of the cells receive and maintain the genes long-term. To select transformants, genes for antibiotic resistance are usually used. We use a gene which confers resistance to the antibiotic, kanamycin. Selection for the transformed cells takes place in a medium containing kanamycin, on which the transformed cells are able to grow and develop into embryos. Normal cells without the newly inserted gene will die on medium with kanamycin, so that the only growth observed should originate from cells with the newly inserted genes.
In our lab, the biolistic process was initially tested for grapevine transformation using embryogenic cell suspensions of 'Chancellor' (supplied by Dr. R.N. Goodman, Missouri). The embryogenic cell suspensions were bombarded with "marker genes" that enable us to track transformation and gene expression. From these cultured plantlets, we have successfully obtained transformed vines expressing foreign genes (work of D. Hébert-Soulé, visiting scientist). One of the genes codes for an enzyme which turns the plant tissue blue when the proper substrate is provided. Visual proof of transformation may be observed when plants are transformed with this gene. Tissues from our transformed vines, now growing in a greenhouse, turn a very dark, blue color when supplied with the substrate. Using common techniques in molecular biology, we have been able to extract DNA from the transformed vines and prove the presence of foreign DNA supplied via the biolistic process.
With our successful 'Chancellor' model system in place, current work focuses on the use of a chitinase-producing gene to confer disease resistance upon important grapevine cultivars. In the laboratory, the chitinase enzyme attacks fungal cell walls and has been shown to inhibit the growth of pathogens that cause Botrytis bunch rot and powdery mildew of grapes. Over the past 2 years, we have been able to produce embryogenic cultures of 'Merlot', 'Chardonnay', 'Pinot noir', 'Concord' and 'Niagara'. Experiments with 'Merlot' , 'Chancellor', and 'Chardonnay' are most advanced, and cultures have been bombarded with the chitinase gene. Recent results indicate that the chitinase gene is expressed in 'Chancellor' and 'Merlot', but it is too early to judge whether the level of disease resistance has been increased. Further experiments are required to obtain plants from these cultures and to judge the effect of this gene on disease resistance.
In the future, it is likely that multiple genes for disease resistance will be inserted simultaneously into important cultivars. There is concern that the product of a single gene will be more readily overcome by a pathogen, and that by pyramiding multiple genes, the resistance will be stable and long lasting. New genes are being sought from grapevines and other organisms. Attempts to create genetic maps of grapevine chromosomes at Cornell University should lead to the isolation of important genes. Finally, any genetically altered vines will have to undergo stringent field testing to assure that, not only is the resistance stable, but that the essential features of the vine and the fruit produced are not altered. For wine grapes, it will be up to the regulatory authorities of the Bureau of Alcohol, Tobacco and Firearms to determine whether wine made from transgenic 'Merlot' may be labeled 'Merlot' on the bottle. There is good precedent for this with 'Pinot noir' in that clones of 'Pinot noir', some of which have likely arisen due to mutation, are permitted to be used in wines labeled with the name 'Pinot noir'.
The final challenge will be to assure a skeptical public of the value of a transgenic grapevine. This technology will help to reduce reliance on pesticides, reduce the cost of production, and permit continued productivity in vineyards hit with harmful virus diseases. In 5-10 years, when transgenic vines and rootstocks become commercially available, the public should have become more accustomed to the consumption and use of transgenic fruits, vegetables, and food and fiber crops. There are already transgenic tomatoes, squash, potatoes and cotton on the market. Improved forms of important grape varieties should not be far behind.
Our research program is supported by dedicated sponsors, including Pebble Beach Winery, Inc. and BARD, the US-Israeli Binational Agricultural Research & Development Fund. We are grateful for this support, as well as past support from the NY Wine & Grape Foundation.
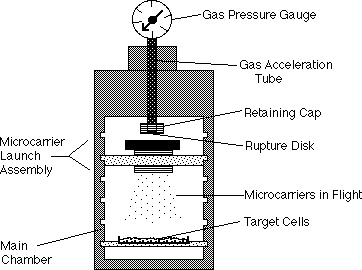
Figure 1. Diagram of the "gene gun" which is used to deliver genes into plant cells. The device is driven by high pressure helium gas. When the rupture disk at the end of the gas acceleration tube is burst, a strong shock wave of gas is released, which in turn launches the microcarriers (minute tungsten particles coated with the desired genes). The microcarriers penetrate the plant cells and the genes are released within. When conditions are optimized, cell injury is minimal and the new genes are maintained by the plant cells long-term. The gene gun was invented by John Sanford, Ed Wolf, and Nelson Allen at Cornell University. The device is being used worldwide to genetically engineer a variety of organisms, including plants, animals, and microorganisms. Medical applications such as gene therapy, are also being tested.
Back to Bruce Reisch's Page.